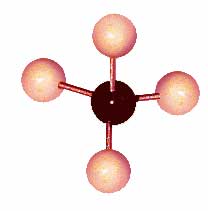
A model of a methane molecule – the simplest of hydrocarbons
A kind of molecule
Hydrocarbons are molecules. They are made out of hydrogen atoms and carbon atoms. Methane is the simplest hydrocarbon molecule. It’s just one carbon atom with four hydrogen atoms stuck to it.
What is hydrogen?
What is carbon?
More about molecules
All our chemistry articles
Are hydrocarbons rare?
Nope. Both carbon and hydrogen were common in nebulae, in exploded stars. So hydrocarbon molecules are very common in space. Many of these molecules are small, like methane. But others are very big. Many hydrocarbons involve hundreds of atoms. Carbon atoms hook together easily. So carbon can easily make long chains of carbon atoms. It’s easy for big molecules to form using carbon as a building block.
What is a nebula?
Bigger hydrocarbons

Lumps of coal
Out in space, the biggest hydrocarbon molecules are amino acids. But here on Earth, those amino acids hooked up with each other to make bigger molecules. We call those protein molecules.
Amino acids
Proteins
One protein molecule can include hundreds of amino acids. They hook together in many different ways, even in the same molecule.
Living cells

A cell dividing into two cells
All living cells are made out of those protein molecules. So all living cells are also made out of hydrocarbons. That includes your body, fish, sweet potatoes, and trees. It includes cheese, and seaweed. All those things are made of hydrocarbon molecules.
The evolution of cells
Anything that was once alive is also made of hydrocarbons. That includes things like oil (made out of plants that died billions of years ago), gasoline and plastic (made out of oil). It also includes rubber, wood, food, alcohol, and many medicines like antibiotics.



[…] and platinum in order to make the catalyst last as long as possible. The reaction is then caused by hydrocarbon molecules breaking […]