Diagram of a sulphur atom
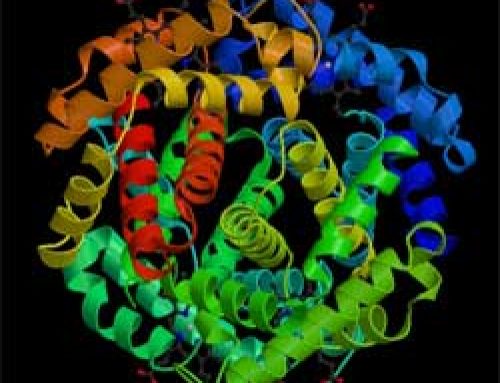
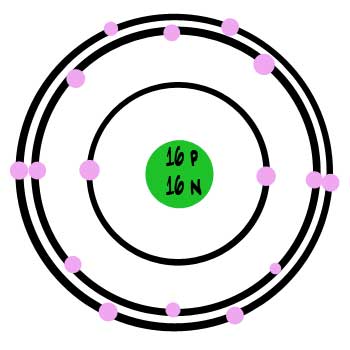
Sulphur atoms fall near the middle of the scale: they are heavier than silicon but lighter than iron. Every atom of sulphur has 16 protons and generally 16 neutrons. Like silicon and iron, all atoms of sulphur are made inside stars that are getting older and have used up most of their fuel but have not yet turned into supernovas. When the star does turn into a supernova, it shoots out the sulphur, and some of it becomes part of planets like Earth. We also know that there’s a lot of sulphur on Io, one of the moons of Jupiter.

Sulphur rocks
Here on Earth, sulphur usually looks like a soft yellow rock. There’s a lot of sulphur in egg yolks, which is part of what makes the yolks yellow, and if you smell a cooked egg yolk it will smell like hydrogen sulfide – a molecule of hydrogen and sulphur atoms. It’s the sulphur that makes rotten eggs smell so bad, too. The reason egg yolks have so much sulphur is that all living cells, both plants and animals, are made partly of sulphur, so when the egg is growing into a new baby, that baby has to use a lot of sulphur to grow.
Where do we get sulphur? Men digging up sulphur from a volcano in Indonesia
Digging up sulfur is hard and dangerous work. Men climb up to the edge of volcanoes and dig up the sulphur there and carry it down from the volcano in baskets.
Learn by doing – Sulphur matches and eggs
More about eggs
Bibliography and further reading about sulphur and chemistry: