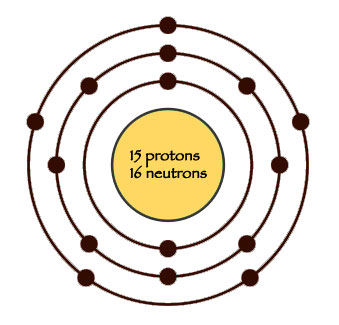
Diagram of a phosphorus atom
Phosphorus atoms have fifteen protons and sixteen neutrons, so they’re just a little lighter than sulphur atoms, in the middle of the range for atoms. Stars make phosphorus when two oxygen atoms get very hot and squash together into one phosphorus atom.
All living things, both plants and animals, are made partly of phosphorus, and they can’t live without it. We need phosphorus to make lipids for cell membranes, and also to make RNA and DNA.

A lighted match
On earth, nearly all of the phosphorus atoms have combined with oxygen atoms to make molecules of phosphate. People use phosphates mainly as fertilizers for plants, but also in matches, where the phosphorus reacts with sulphur when you strike a match to make the flame. (Phosphorus itself is poisonous to people, so today matches use a different version of phosphorus that’s safer.)
Learn by doing – Phosphorus and Matches
Bibliography and further reading about atoms and phosphorus:




